Coiling Procedures: some new optimization perspectives
Coiling procedures are no longer to be defined. For more than 30 years, they have been the basis for the extension of the endovascular approach for the treatment of intracranial aneurysms, and remains the most frequently used procedure.(1,2)
The efficacy and safety of this procedure have also been widely documented. As a major example, the International Subarachnoid Aneurysm Trial (ISAT) demonstrated that in patients considered eligible for endovascular coiling and neurosurgical clipping, the clinical outcome at 1 year after aneurysm rupture was significantly better with endovascular coiling. Similarly, after longer follow-up, the probability of disability-free survival was significantly greater in the endovascular group than in the neurosurgical group.(3,4)
That said, what room is left for improvement and innovation?
Here is the place where new technologies such as simulation can give us perspectives!
Let’s deep dive into the experience we have with coils.
Aneurysm recurrence after coiling
Many studies have been designed to evaluate the efficacy of coiling treatment. The recurrence or recanalization rate for patients treated with coils alone is 28.9%(5) compared to around 14% with flow diversion(6,7) and 16.4% of inadequate occlusion with intrasaccular flow disrupters like the Woven EndoBridge™(8). The associated potential dangers of aneurysm recanalization are bleeding and/or retreatment: morbidity and mortality of retreatment ranging from 6 to 11%.(9)
Recanalization can occur by a variety of mechanisms including(5): Coil compaction and migration, or Aneurysm growth.
Factors correlated to the risk of recurrence
If we push the analysis further, we can note that many factors have been associated with an increased risk of recurrence. Among these, some are physio pathological and specific to the aneurysm (5,10,11):
- Aneurysm rupture
- Aneurysm size > 10 mm
- Wide-necked aneurysms
- Location of the aneurysm
However, other factors directly related to the coiling procedure have been identified and could be optimized:
VER (Volume Embolization ratio) or PD (Packing Density)
The VER is obtained by dividing the volume of coils deployed by the total volume of the aneurysm.
According to literature, the optimal value to reach at the end of the procedure after all coils have been deployed varies between 24 and 33%.(12–17) By reaching these values, the recurrence rate is significantly reduced.
Framing Coil or First Coil
The framing coil is considered the most important coil of the procedure. As for the packing density, the VER cut-off to be reached, for the first coil alone, is statistically >10% in order to decrease the risk of recurrence.(12,18)
However, to increase the chances of achieving a satisfactory total VER and better results, the VER value of the framing coil to aim for is ideally between 17.5 and 20%.(12)
fCsize
This is where Sim&Cure’s technologies come in. Through new features, the FCsize™ simulations give more insights to the physician to improve these parameters.
With the support of the Sim&Size™ medical device software, including the FCsize™ module, the physician can simulate several coils devices and test different treatment scenarios in real time.
In a few clicks, the simulation enables to focus first on the framing coil: based on its apposition within the aneurysm and the VER value displayed, the physician will choose the most suitable framing coil.
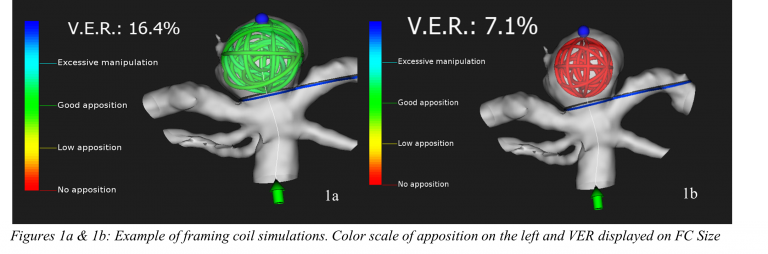
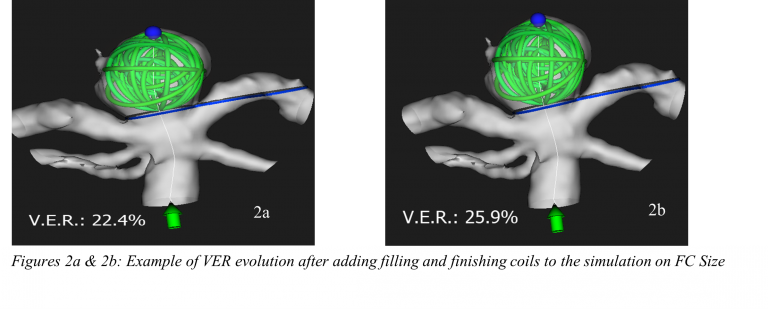
As mentioned above, this first choice is critical and can be delicate. Consequently, multiple first coil simulations can be run simultaneously. Then, the physician can add the filling and finishing coils to the simulations, while observing the evolution of the VER, and compare the potential choices he has.
Also compatible with the STsize™ and FDsize™ simulation modules, it is possible to evaluate the impact of stents and flow diverters in the specific patient anatomy.
These features allow to support the planning of the entire procedure prior to the intervention in order to secure the patient’s treatment. With the emergence of sizing technologies based on artificial intelligence and digital twin, the choice of coils and their size can be optimized before the deployment.
All these possibilities make FCsize™ a singular tool on the market. Among the first experiences our users had, these new configurations already impact the clinical practice, as for example, by “avoiding the deployment of unnecessary coils, also preventing an additional risk for the patient” as mentioned by Pr. Costalat.
By combining patient and device specific simulation, the usage of the Sim&Size™ software aim to reduce the number of manipulations and additional materials deployed (19,20). In the same way, the usage of such tool could further homogenize and standardize the management of patients with brain aneurysms, based on scientific evidence and three-dimensional analysis.
Sim&Size™ is a medical device regulated health product manufactured by Sim&Cure. According to the European regulation, Sim&Size™ software is a Class Ila medical device, it carries, under this regulation, the CE marking n°0459. The Sim&Size software is a medical device intended to provide a 3D view of the final deployment of implants. It uses an actual image of the patient produced by 3D rotational angiography. It offers clinicians the possibility of simulating deployment of implantable medical devices (IMD) in the artery to be treated and predicts the final position (length, diameter, form) and the extent of apposition of the implant.
Références
1. Brinjikji W, Rabinstein AA, Nasr DM, Lanzino G, Kallmes DF, Cloft HJ. Better Outcomes with Treatment by Coiling Relative to Clipping of Unruptured Intracranial Aneurysms in the United States, 2001–2008. Am J Neuroradiol. 2011 Jun;32(6):1071–5.
2. Tawk RG, Hasan TF, D’Souza CE, Peel JB, Freeman WD. Diagnosis and Treatment of Unruptured Intracranial Aneurysms and Aneurysmal Subarachnoid Hemorrhage. Mayo Clin Proc. 2021 Jul;96(7):1970–2000.
3. Molyneux AJ, Kerr RSC, Yu LM, Clarke M, Sneade M, Yarnold JA, et al. International subarachnoid aneurysm trial (ISAT) of neurosurgical clipping versus endovascular coiling in 2143 patients with ruptured intracranial aneurysms: a randomised comparison of effects on survival, dependency, seizures, rebleeding, subgroups, and aneurysm occlusion. 2005;366:9.
4. Molyneux AJ, Birks J, Clarke A, Sneade M, Kerr RSC. The durability of endovascular coiling versus neurosurgical clipping of ruptured cerebral aneurysms: 18 year follow-up of the UK cohort of the International Subarachnoid Aneurysm Trial (ISAT). The Lancet. 2015 Feb;385(9969):691–7.
5. Pierot L, Barbe C, Thierry A, Bala F, Eugene F, Cognard C, et al. Patient and aneurysm factors associated with aneurysm recanalization after coiling. J NeuroInterventional Surg. 2021 Nov 5;neurintsurg-2021-017972.
6. Bender MT, Colby GP, Lin LM, Jiang B, Westbroek EM, Xu R, et al. Predictors of cerebral aneurysm persistence and occlusion after flow diversion: a single-institution series of 445 cases with angiographic follow-up. J Neurosurg. 2018 Mar;1–9.
7. Dmytriw AA, Phan K, Salem MM, Adeeb N, Moore JM, Griessenauer CJ, et al. The Pipeline Embolization Device: Changes in Practice and Reduction of Complications in the Treatment of Anterior Circulation Aneurysms in a Multicenter Cohort. Neurosurgery [Internet]. 2020;86(2). Available from: https://journals.lww.com/neurosurgery/Fulltext/2020/02000/The_Pipeline_Embolization_Device__Changes_in.12.aspx
8. Pierot L, Szikora I, Barreau X, Holtmannspoetter M, Spelle L, Herbreteau D, et al. Aneurysm treatment with WEB in the cumulative population of two prospective, multicenter series: 3-year follow-up. J NeuroInterventional Surg. 2021 Apr;13(4):363–8.
9. Muskens IS, Hertgers O, Lycklama à Nijeholt GJ, Broekman MLD, Moojen WA. Outcomes of Retreatment for Intracranial Aneurysms — A Meta-Analysis. Neurosurgery [Internet]. 2019;85(6). Available from: https://journals.lww.com/neurosurgery/Fulltext/2019/12000/Outcomes_of_Retreatment_for_Intracranial_Aneurysms.3.aspx
10. Han Y, Liu J, Tian Z, Lv M, Yang X, Wu Z, et al. Factors affecting recurrence and management of recurrent cerebral aneurysms after initial coiling. Interv Neuroradiol. 2020 Jun 1;26(3):300–8.
11. Damiano RJ, Tutino VM, Paliwal N, Patel TR, Waqas M, Levy EI, et al. Aneurysm characteristics, coil packing, and post-coiling hemodynamics affect long-term treatment outcome. J NeuroInterventional Surg. 2020 Jul 1;12(7):706.
12. Neki H, Kohyama S, Otsuka T, Yonezawa A, Ishihara S, Yamane F. Optimal first coil selection to avoid aneurysmal recanalization in endovascular intracranial aneurysmal coiling. J NeuroInterventional Surg. 2018 Jan 1;10(1):50.
13. Slob MJ, Sluzewski M, van Rooij WJ. The relation between packing and reopening in coiled intracranial aneurysms: a prospective study. Neuroradiology. 2005 Dec 1;47(12):942–5.
14. Kawanabe Y, Sadato A, Taki W, Hashimoto N. Endovascular Occlusion of Intracranial Aneurysms with Guglielmi Detachable Coils: Correlation Between Coil Packing Density and Coil Compaction. Acta Neurochir (Wien). 2001 Jun 1;143(5):451–5.
15. Sluzewski M, van Rooij WJ, Slob MJ, Bescós JO, Slump CH, Wijnalda D. Relation between Aneurysm Volume, Packing, and Compaction in 145 Cerebral Aneurysms Treated with Coils. Radiology. 2004;231(3):653–8.
16. Tamatani S, Ito Y, Abe H, Koike T, Takeuchi S, Tanaka R. Evaluation of the Stability of Aneurysms after Embolization Using Detachable Coils: Correlation between Stability of Aneurysms and Embolized Volume of Aneurysms. 2002;6.
17. YAGI K, SATOH K, SATOMI J, MATSUBARA S, NAGAHIRO S. Evaluation of Aneurysm Stability After Endovascular Embolization With Guglielmi Detachable Coils: Correlation Between Long-Term Stability and Volume Embolization Ratio. Neurol Med Chir (Tokyo). 2005;45(11):561–6.
18. Sousa EB, Brandão LF de S, Tavares CB, Brito JNP de O, Kessler IM. Importance of the First Coil in the Embolization of Intracranial Aneurysms. Clin Neuroradiol. 2019 Dec 1;29(4):733–40.
19. Piergallini L, et al. Virtual simulation with Sim&Size software for Pipeline Flex Embolization: evaluation of the technical and clinical impact. J NeuroIntervent Surg 2020; 0:1–6. doi:10.1136/neurintsurg-2020-015813
20. Cagnazzo F, Marnat G, Ferreira I, et al. Comparison of Woven EndoBridge device sizing with conventional measurements and virtual simulation using the Sim&Size software: a multicenter experience. J NeuroIntervent Surg Epub ahead of print: [23 December 2020]. doi:10.1136/ neurintsurg-2020-017060
